Atomic Structure 1
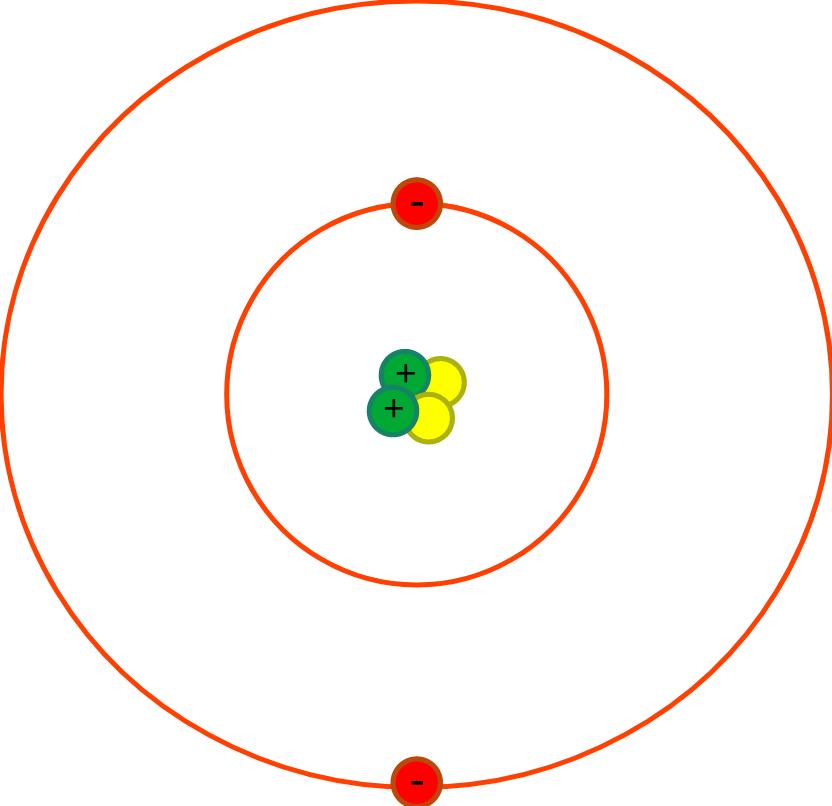
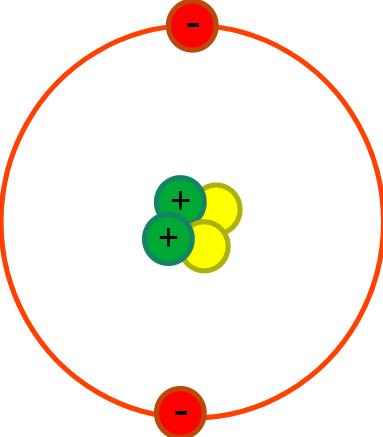
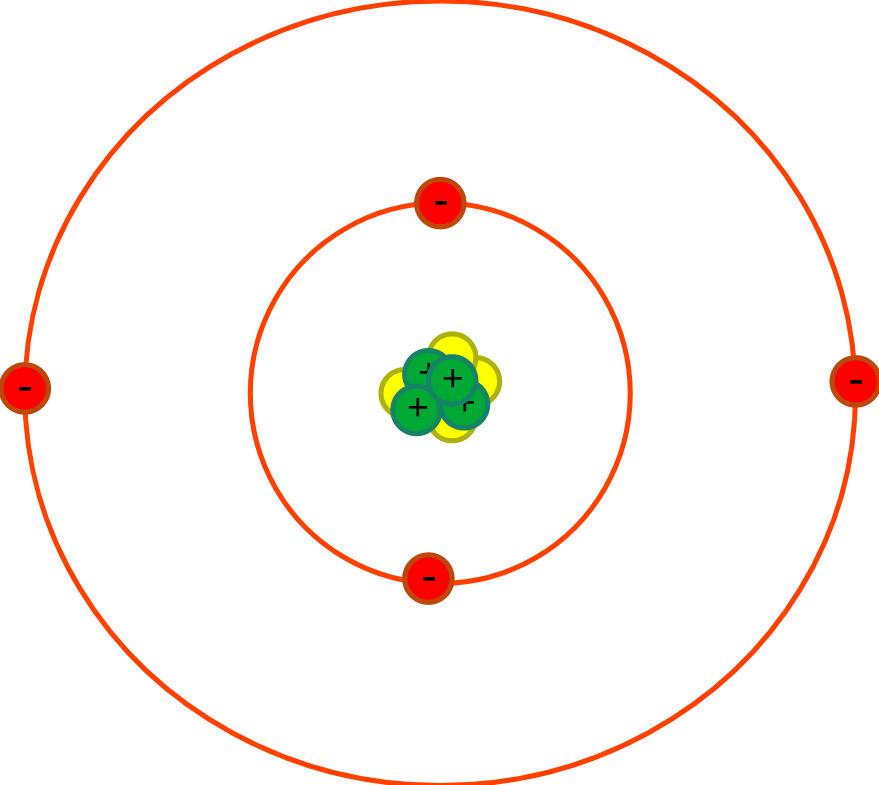
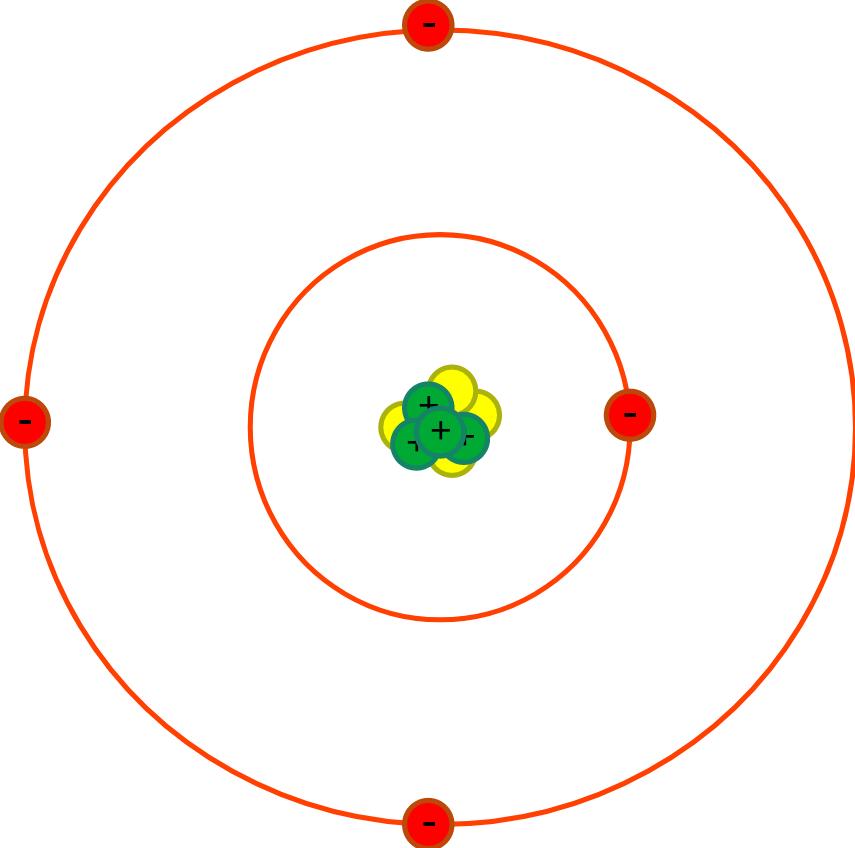
| First | First | Last | Quiz Score Total | 1a) Which of the following shows the typical radius of an atom? | 1b) The radius of the nucleus is less than 1 / 10000th the radius of the atom. Which of the following shows the radius of the nucleus? | 1ci) What is the part labelled A? | 1cii) What is the part labelled B? | 1ciii) What is the part labelled C? | 1ciiii) What is the part labelled D? | 1di) What charge do protons have? | 1dii) What charge do electrons have? | 1diii) What charge do neutrons have? | 1e) Why does the nucleus has an overall positive charge? | 1f) Why does the charge on the nucleus not depend on the number of neutrons? | 1gi) What is happening to the Electromagnetic Radiation in the diagram above? | 1gii) What diagram below shows the energy level at its highest? | 1hi) What is happening to the Electromagnetic Radiation in the diagram above? | 1hii) What diagram below shows the energy level at its highest? | Quiz Percentage Grade |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ppefmh | Ed | Garstin | 9/15 |
|
|
|
|
|
|
|
|
|
|
|
|
| 60% | ||
| First | First | Last | Quiz Score Total | 1a) Which of the following shows the typical radius of an atom? | 1b) The radius of the nucleus is less than 1 / 10000th the radius of the atom. Which of the following shows the radius of the nucleus? | 1ci) What is the part labelled A? | 1cii) What is the part labelled B? | 1ciii) What is the part labelled C? | 1ciiii) What is the part labelled D? | 1di) What charge do protons have? | 1dii) What charge do electrons have? | 1diii) What charge do neutrons have? | 1e) Why does the nucleus has an overall positive charge? | 1f) Why does the charge on the nucleus not depend on the number of neutrons? | 1gi) What is happening to the Electromagnetic Radiation in the diagram above? | 1gii) What diagram below shows the energy level at its highest? | 1hi) What is happening to the Electromagnetic Radiation in the diagram above? | 1hii) What diagram below shows the energy level at its highest? | Quiz Percentage Grade |